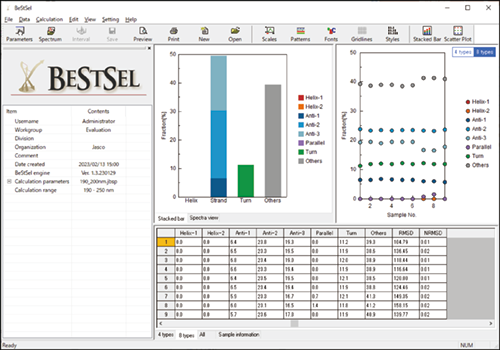
Spectra Manager BeStSel program
While many academic researchers use the BeStSel web server, pharmaceutical companies are unable to use their proprietary data on the site, and seek a development environment where the data can be analyzed offline. Addressing this need, an offline BeStSel software in Spectra Manager™ 2.5 (Spectra Manager BeStSel) was jointly developed, and enables seamless analysis in an offline capacity. Furthermore, Spectra Manager™ 2.5 CFR BeStSel was also developed, ensuring compatibility with GxP and satisfying computer system validation (CSV), electronic record/electronic signature (ER/ES) and data integrity (DI) for practicing ALCOA+ principles.
If you have any question about this product or quotation request, please kindly contact our JASCO regional sales representatives.
Application
Thermal denaturation analysis of monoclonal antibody
The advantage of BeStSel is not only its high accuracy in analyzing β-structure-rich-proteins, but also its feature of being able to calculate the fraction of eight secondary structure elements, including three types of twisting in antiparallel β-sheets.
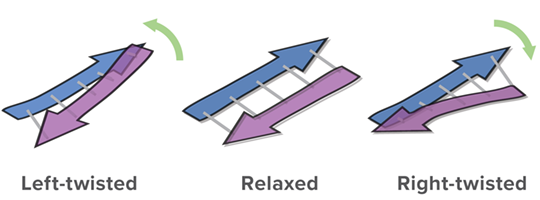
Twisting of β-strand
As assessment examples by using BeStSel, the detailed secondary structure changes as a function of temperature were monitored. There is a significant difference between Herceptin® and h-IgG in changes in the distorted α-helix, left-twisted β-strand, relaxed β-strand and right-twisted β-strand. At 30 °C, h-IgG and Herceptin® have a similar secondary structure, but it was found that they have different denaturation mechanisms by using BeStSel. BeStSel is shown to be useful in understanding the detailed mechanisms of structure formation in proteins, including therapeutic antibodies.
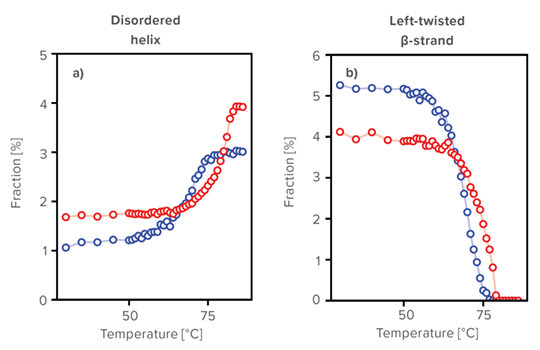
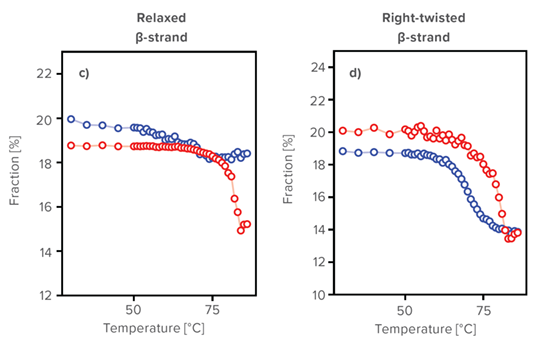
Results of secondary structure prediction using BeStSel
Predicted secondary structure fraction of a) disordered Helix, b) left-twisted, c) relaxed, and d) right-twisted β-strand of Herceptin® (red) and human IgG (blue).
If you are interested in the on-line version of BeStSel software, please go to the following BeStSel web site at ELTE Eötvös Loránd University.

