Quantification of coumarin
Legally, a small amount of coumarin (determined by country) must be added to kerosene to distinguish it from diesel oil. To prevent the production of illegal diesel oil, a standardized analytical method for the quantitative determination of the coumarin content using fluorescence spectroscopy has been established (Japan Petroleum Institute Standard: JPI-5S-71-2010 ). The coumarin is first hydrolyzed in an alkaline solution and is then isomerized to a "trans" form by UV irradiation (Fig. 11).
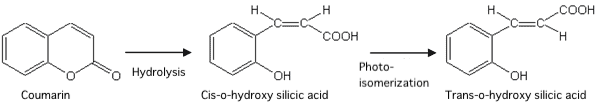
Fig. 11 Hydrolysis and photoisomerization of coumarin.
The resulting samples were then analyzed using fluorescence spectroscopy. Calibration curves obtained from samples containing mixtures of coumarin-free and coumarin-containing oil with mixing ratios of 0, 20, 40, and 80% exhibited a correlation coefficient of 0.9996 (Fig. 12). The standard error (σ) between the measured mixing ratio and the actual mixing ratio was 0.65%. The detection limit for the mixing ratio was 2.1% and the lower quantification limit was 6.5%.
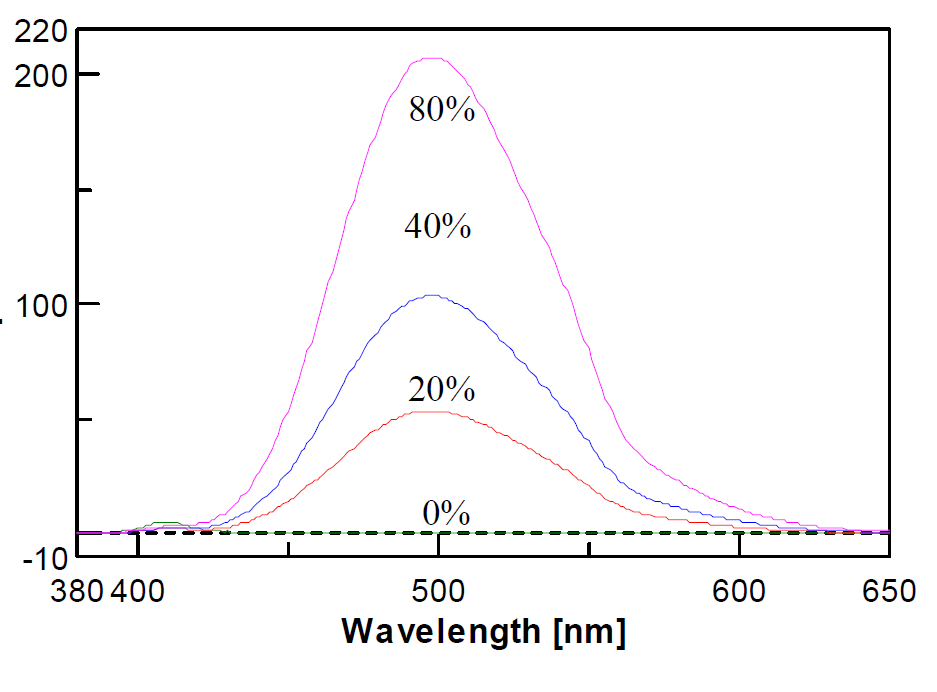
Fig. 12-1 Fluorescence spectra
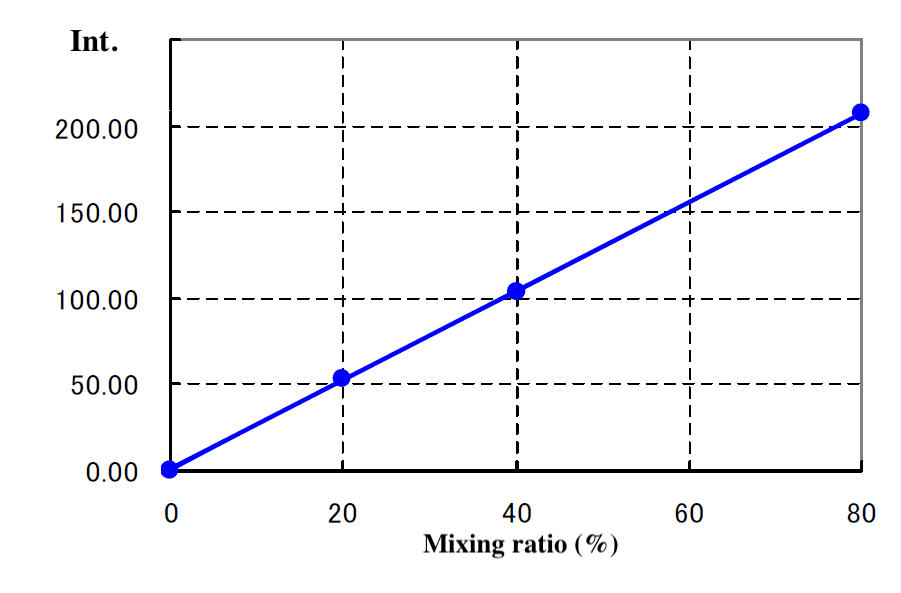
Fig. 12-2 Calibration curve for mixing ratio based on coumarin detection
Reflectance measurements using spectrofluorometer
It is well known that it is difficult to perform reflectance measurements on fluorescent samples using a UV/Vis spectrometer. This is because the detected light contains a fluorescence component so that the true absorption spectrum cannot be obtained. In contrast, using a spectrofluorometer and synchronous scanning so that the wavelength difference between Ex and Em is zero, the true transmission and reflection spectra can be obtained. Fig. 13 shows the reflection spectrum of a fluorescent red card. When the sample does not emit fluorescence, the spectrum measured with a spectrofluorometer matches that measured with a spectrometer. This is an example of the use of a spectrofluorometer for spectroscopic measurement of a fluorescent sample.
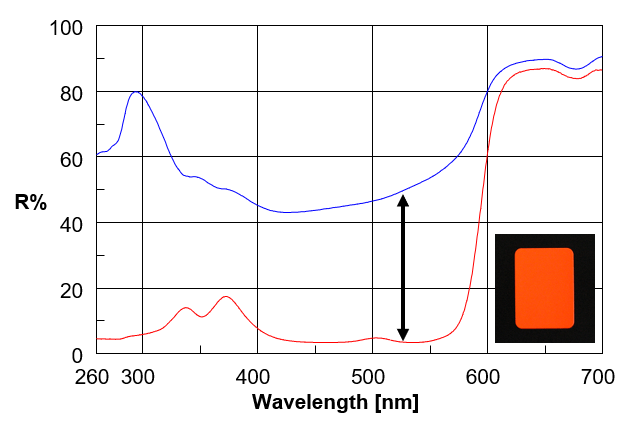
Fig. 13 Reflectance measurement of fluorescent red card (Blue: Reflection spectrum obtained using UV/Vis spectrometer, Red: Reflection spectrum obtained using spectrofluorometer)
Phosphorescence measurement
Phosphorescence due to intersystem crossing has a longer lifetime than fluorescence, and can be measured by momentarily interrupting the excitation light using a shutter. If oxygen quenching or thermal inactivation by a solvent is likely to occur, the sample can be cooled to liquid nitrogen temperature and frozen (Fig. 14). In recent years, research on materials that emit phosphorescence at room temperature, such as organic EL pigments, is also progressing.
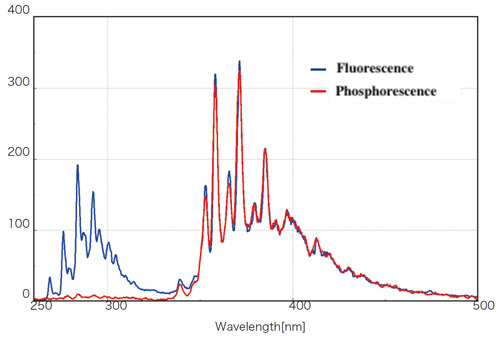
Fig. 14 Phosphorescence and fluorescence spectra of benzene
Related Posts:
 Quantitative Determination of Low-concentration…
Quantitative Determination of Low-concentration… Quantification Method for Oil and Grease in Water…
Quantification Method for Oil and Grease in Water… Determination of Absolute Configuration of Molecules…
Determination of Absolute Configuration of Molecules… Two-dimensional Correlation Spectroscopy for…
Two-dimensional Correlation Spectroscopy for… Can the JASCO Palmtop Raman spectrometer perform a…
Can the JASCO Palmtop Raman spectrometer perform a…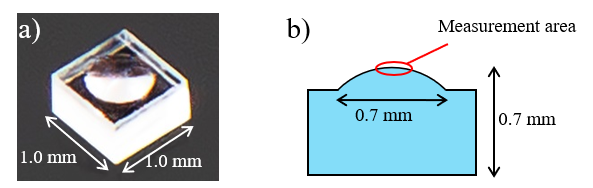 Evaluation of Reflectance of Microlens Surface using…
Evaluation of Reflectance of Microlens Surface using…
