Introduction
CD spectroscopy is one of the leading techniques in protein structure analysis, with fluorescence spectroscopy and fluorescence anisotropy both providing complementary information. While CD spectra provide information regarding the secondary structure of proteins, fluorescence spectra and anisotropy provide information about the local environment surrounding the fluorophores. In particular, fluorescence anisotropy provides additional information about the rotational movement of these fluorophores which cannot be obtained by fluorescence spectroscopy alone.
The JASCO J-1500 CD spectrometer with dedicated fluorescence anisotropy kit enables to measure spectrum for fluorescence anisotropy and to know rotational movement in proteins.
This article describes the changes in fluorescence anisotropy measurements during the denaturation of α-lactalbumin by guanadinium hydrochloride (GuHCl)1).

J-1500 CD spectrometer
Experimental
Measurement parameters
Data acquisition interval: 0.1 nm
Excitation bandwidth: 7 nm
Accumulation: 4 times
D.I.T.: 2 sec
Scan speed: 100 nm
Keywords
Circular dichroism, fluorescence, anisotropy, α-lactalbumin, GuHCl, secondary structure, denaturation, biochemistry
Results
The fluorescence anisotropy spectra of both the native-state α-lactalbumin in H2O and the unfolded α-lactalbumin in 3.4 M GuHCl are shown in Figure 1. Both spectra show a peak maxima at 267 nm and peak minima at 283 and 291 nm. All three peaks result from the tryptophan residue of the protein2). Figure 1 illustrates that the denaturation of α-lactalbumin clearly involves the decreases in fluorescence anisotropy, indicating a more freely rotating tryptophan residue.
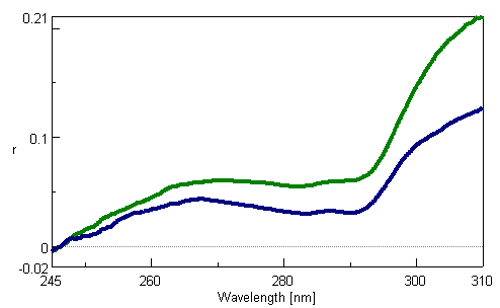
Figure 1. Fluorescence anisotropy spectra of the denaturation of α-lactalbumin by GuHCl. The green line indicates 0.02 mg/mL α-lactalbumin, 0.1 mM EDTA in the absence of GuHCl, and the blue line is 0.02 mg/mL α-lactalbumin, 0.1 mM EDTA in 3.4 M GuHCl.
References
1. Canet, D., Doering, K., Dobson, C. M., and Y. Dupont, Biophysical Journal (2001), 80, 1996-2003.
2. J. R. Lakowicz, Principles of Fluorescence Spectroscopy, New York, 446-487.