Separation principle
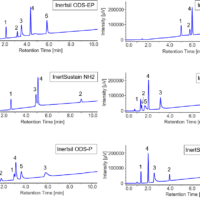
In HPLC, individual components are separated using a column, based on the difference in the degree of interaction between the sample components and the column. Components with a low degree of interaction with the column are eluted first. These interactions include adsorption, hydrophilic interactions, hydrophobic interactions, electroaffinity, penetration and exclusion (Fig. 6).
Fig. 6 Separation method
Column types and separation modes
As shown in Table 2, there are various types of columns and separation modes that can be used, and the optimum choice depends on the nature of the sample and the analysis that is required. When an organic solvent is used as the mobile phase, a normal-phase column (mainly silica gel) can separate and analyze samples composed of fat-soluble components based on adsorption. When a water/methanol solvent is used as the mobile phase, separation can be achieved based on hydrophobic interactions in reverse-phase mode. GPC columns separate sample components based on their molecular size using pores. Ion exchange columns separate ion components based on electrical affinity.
Table 2 Column types and separation modes
| Mode | Stationary phase | Mobile phase | Interaction | Features |
| Normal phase | Silica gel | Organic solvent | Adsorption | Separation of fat-soluble components |
| Reversed phase | Silica C18 (ODS) | Water / MeOH | Hydrophobic | The most commonly used method |
| GPC (non-aqueous) | Polymer | Organic solvent | Gel permeation | Molecular weight distribution measurement |
| GFC (aqueous) | Hydrophilic polymer | Buffer | Gel permeation | Biopolymer separation |
| Ion exchange | Ion exchanger | Buffer | Electric affinity | Separation of ionic components |
Normal phase vs. reverse phase
Normal-phase chromatography and reverse-phase chromatography are completely different methods. In normal-phase chromatography, a low-polarity solvent is passed through a high-polarity column and the low-polarity components are eluted first. In reverse-phase chromatography, which is the most commonly employed technique, short-carbon-chain compounds are eluted first.
Isocratic elution vs. gradient elution
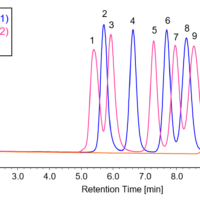
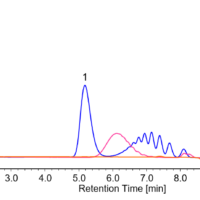
In reverse-phase chromatography and ion-exchange chromatography, gradient elution may be used to improve the separation and reduce the measurement time. As an example, chlorogenic acid and rutin can be separated using an ODS column and a methanol/1% acetic acid solution.
First, let’s look at an analysis example that was performed without changing the composition ratio of the solvent. In the case of a methanol/1% acetic acid ratio of 40/60, component A could not be separated well (Fig. 7, upper left). In the case of a methanol/1% acetic acid ratio of 30/70, separation was completed but it took a long time (Fig. 7, lower left). However, by applying a gradient from a methanol/1% acetic acid ratio of =30/70 to 45/55 (changing the concentration), the retention of the column was strengthened, and component A was reliably separated and eluted (Fig. 7, right).
Fig. 7 Isocratic elution (left) vs. gradient elution (right)