Introduction
Measuring IR spectra under temperature control is an effective method to study denaturation of DNA and protein, and enzymatic kinetic reaction. Oxidative degradation monitoring by IR measurement of unsaturated fatty acid contained in fat and oil is one of the remarkable examples of this application.

FT/IR-4600 and ATR PRO670H-S
There are two different types of unsaturated fatty acid such as Cis-type and Trans-type, and Cis-type fatty acid is contained mainly in natural oil and Trans-type, in hydrogenated oil in food. It is well known that Cis-type reacts to form peroxide lipids through oxidative degradation by Radical and isomerizes to Trans-type as shown in Fig. 1. In this report, the commercial vegetable oil sample heated up to 180 degree C was measured by using heating single ATR accessory with FTIR and oxidative degradation process was analyzed.
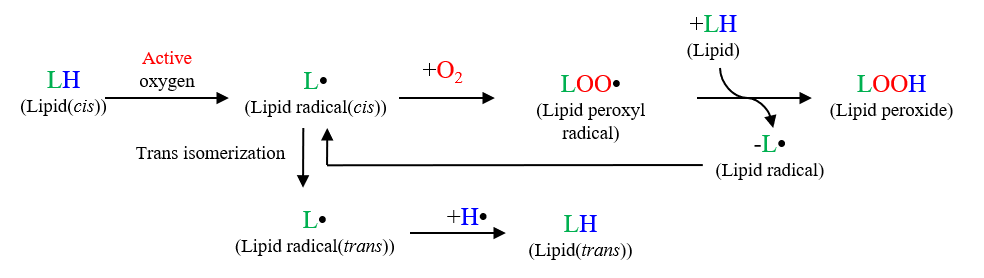
Fig. 1 Reaction mechanism of oxidation degradation of the Lipid
Experimental
There are two different types of unsaturated fatty acid such as Cis-type and Trans-type, and Cis-type fatty acid has specific absorption peak at 3010 cm-1 and Trans-type, at 966 cm-1.
It is possible to evaluate oxidative degradation process of unsaturated fatty acid in oil by monitoring IR peaks at 3010 cm-1 and 966 cm-1, since Cis – Trans transformation will be resulted in the degradation process. Under similar to the actual cooking environment, the sample was heated up to 180 degree C and two absorbance peaks attributed to Cis and Trans fatty acid were measured by interval measurement program, and oxidative degradation process was evaluated by the changes of two peak intensities with time. The same measurement was also implemented under the environment purged by N2 gas.
<Measurement condition>
Main unit: FT/IR-6600 with Heating Single reflectance ATR (ATR PRO670H-S)

Detector: DLATGS
Resolution: 4 cm-1
Accumulation: 64 times
Temperature: 180 degree C
Apodization function: Cosine
Measurement method: ATR (Crystal: Diamond*)
Sample compartment condition: Air environmental condition or N2 purged condition
*The high temperature type prism was used in this measurement.
Results
Fig. 3 shows spectral data of time interval measurement for the oil sample heated up to 180 degree C under air environmental condition. It is seen that peak at 3010 cm-1 attributed to Cis-type is getting smaller with time. On the other hand, peak at 966 cm-1 attributed to Trans-type is getting larger with time. In Fig. 4 the spectral data of the same interval measurement but under N2 purged condition are shown, clearly indicating that there is no change of peaks with time. It can be said that Cis-Trans transformation did not occurr, proving that oxidative degradation of oil did not happen.
Fig. 5 and Fig. 6 illustrates that the changes of peak area with time at 3010 cm-1 and at 966 cm-1 are plotted respectively by using the interval analysis program. It is evident that the peak area for Cis-type at 3010 cm-1 is getting smaller with time gradually under air environment, but the peak area for Trans-type at 966 cm-1 is getting larger with time for the initial 30 minutes and then saturated gradually. By comparing the change of peak area for Cis-type and that for Trans-type with time under air environment shown in Fig. 5 and Fig. 6, it is assumed that after 30 minutes, the reaction to generate lipid peroxide becomes more dominant than the Cis-Transformation as in the reaction process shown in Fig.1 As seen above, it is proved that the FTIR measurement method using a heating single reflection ATR is very effective to monitor and evaluate the reaction process accompanied by thermal changes. It is expected that this method will be expanded to be applied for evaluation of thermodynamic reaction and for measurement under the condition of Radical capture agent added.
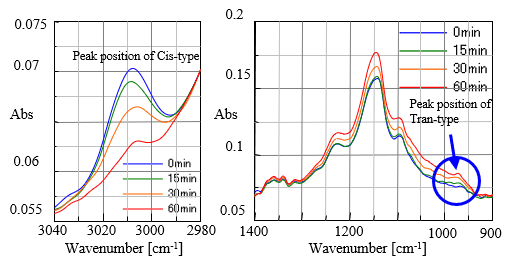
Fig. 3 Spectra under air environmental condition
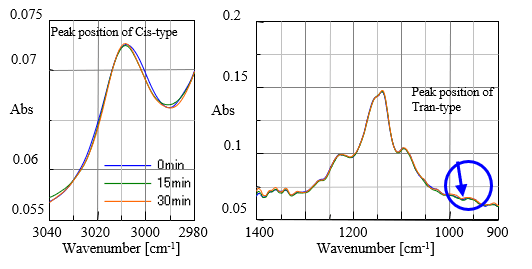
Fig. 4 Spectra under N2 purged condition
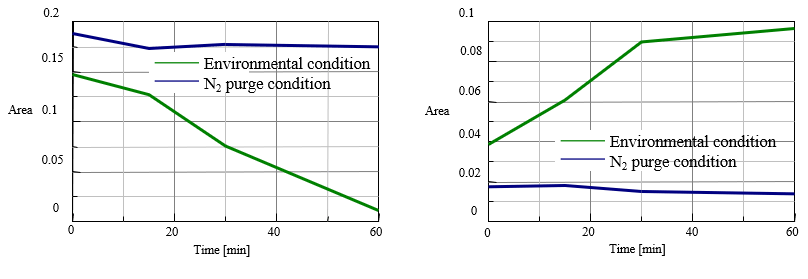
Fig. 5 Change of peak area (left: cis (at 3010 cm-1), right: trans (at 966 cm-1))