Introduction
Peptide mapping is a standard testing method for biomedicines. This method requires HPLC separation of peptide segments digested using enzymes or chemicals and is an internationally harmonized method described by the U.S. Pharmacopoeia (USP), the European Pharmacopoeia (EP)and the Japanese Pharmacopoeia (JP).
The target protein and a standard protein are digested using the same protocol. A direct comparison of the peptide map of the target protein can be made with the standard protein so that the expression of recombinant proteins can be assessed, and this method is applied to the quality assurance.
This article shows the peptide mapping of tryptic digests of BSA by using of UHPLC system.

LC-4000 UHPLC system
Experimental
Chromatographic condition
Column: ZORBAX SB-C18 (2.1 mmID x 150 mmL, 1.8 µm)
Trapcolumn: ODS (2.0 mmID x 10 mmL, 5 µm)
Eluent A: 0.05% Formic acid/Acetonitrile (97/3)
Eluent B: 0.05% Formic acid/Acetonitrile (40/60)
Gradient condition: (A/B), 0 min(100/0) -> 10, 60, 120 min (20/80)
Flow rate: 0.2 mL/min
Column temp.: 40ºC
Wavelength: 215 nm
Injection volume: 5 µL
Standard sample: 1.2 µg/µL BSA tryptic digest
Sample Preparation
1. Dissolve 10 mg of BSA in 1.5 mL of solvent (6 M Urea: 0.1M NH4HCO3=1:4).
2. Add 200 μl of 1% of trypsin in 0.003N HCl to BSA solution to a ratio of BSA solution to 1% trypsin at 5:1 (w/w).
3. Digest and incubate at 37ºC for 15 hours.
4. Perform ultrafiltration using model Ultrafree C3 UFC3LGC00 (10,000MWCO) membrane.
5. Dilute 5 times with mobile phase A.
Keywords
peptide mapping, BSA, tryptic digestion, 1.8 µm, C18 column, PDA detector
Results
Chromatograms of tryptic digests are shown in figure 1. Depending on the gradient profiles, the number of eluted peak varies: 93 peaks in a 10 minute gradient,139 peaks in a 60 minute gradient and 125 peaks in a 120 minute gradient.
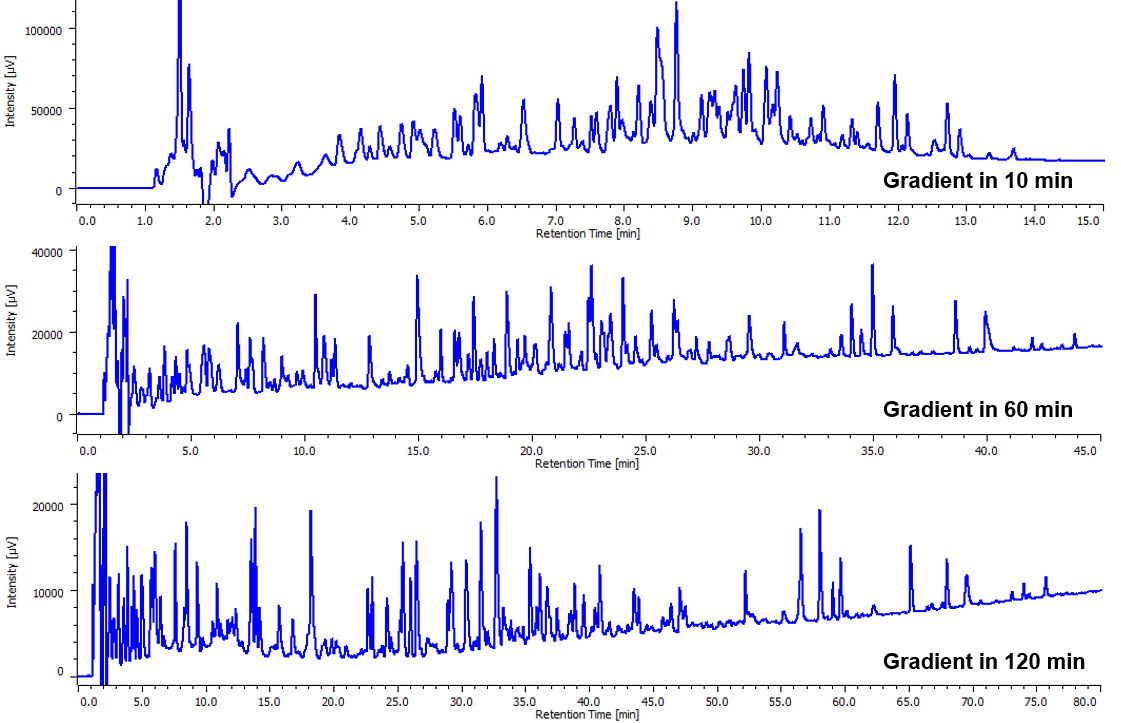
Figure 1. Chromatogram of tryptic digests