Introduction
Effective 22 July, 2019, the revised RoHS directive (EU) 2015/863 restricted the use of four phthalates (see Fig.1) in in children’s toy and mechanical parts. This new directive is very strict, especially regarding the three of the phthalates in toys (DEHP, BBP and DBP); their total concentration should be less than 1000 ppm (0.1%). The recommended method for evaluation phthalates is either GC/MS or Py/TD-GC/MS as used in IEC 62321-8. However, these instruments are complicated to set up and operate, and simpler, cheaper alternative is preferred. JASCO has collaborated with “Kankyo Assist Co., Ltd” to develop a simplified evaluation method using FTIR. This application note offers a detailed evaluation procedure for the controlled phthalates.
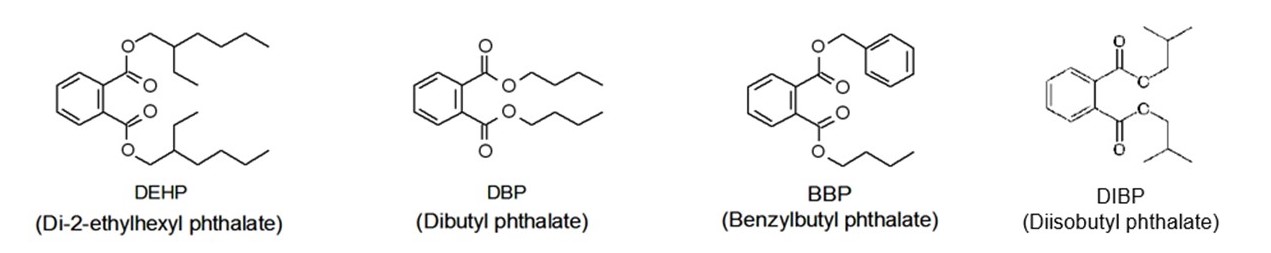
Fig.1 Structures of the four phthalates (restricted by RoHS)
Experimental
Extraction-Condensation method
Conventionally, an FTIR spectroscopic method with ATR method has been used for evaluating phthalates (App Note: 100-AT-0230). Since the ATR method does not require complicated sampling, it offers fast and easy sample preparation and measurement. However, the ATR method does not have sufficient sensitivity to reach the 1000 ppm (0.1%) levels required by the new directive. Therefore, JASCO has developed a new high-sensitivity Extraction-Condensation method.
Using this method phthalates are solvent extracted from the sample, and then made into a thin film for measurement (Fig.2). The LOQ (limit of quantitation) of this method is approximately 700 ppm, and the LOD (limit of detections) is approximately 300 ppm*1). If phthalates are not detected using this method, it can be concluded that the revised RoHS directive has been met. Since the measurement time is approx. 40 min./sample, it is estimated that more than 40 samples/day can be analyzed, if multiple samples are extracted at the same time.
An extraction solvent should be used that dissolves the phthalates without dissolving the resin in the substrate. In the case of PVC resin, a mixed organic solvent (acetone : hexane = 3 : 7) was used. The extracted solution is dropped onto a custom-made substrate*2) , which allows a uniform thin film of the sample to be made, and then the solvent is evaporated. The spectrum of the substrate is measured using an IR reflection-absorption method.
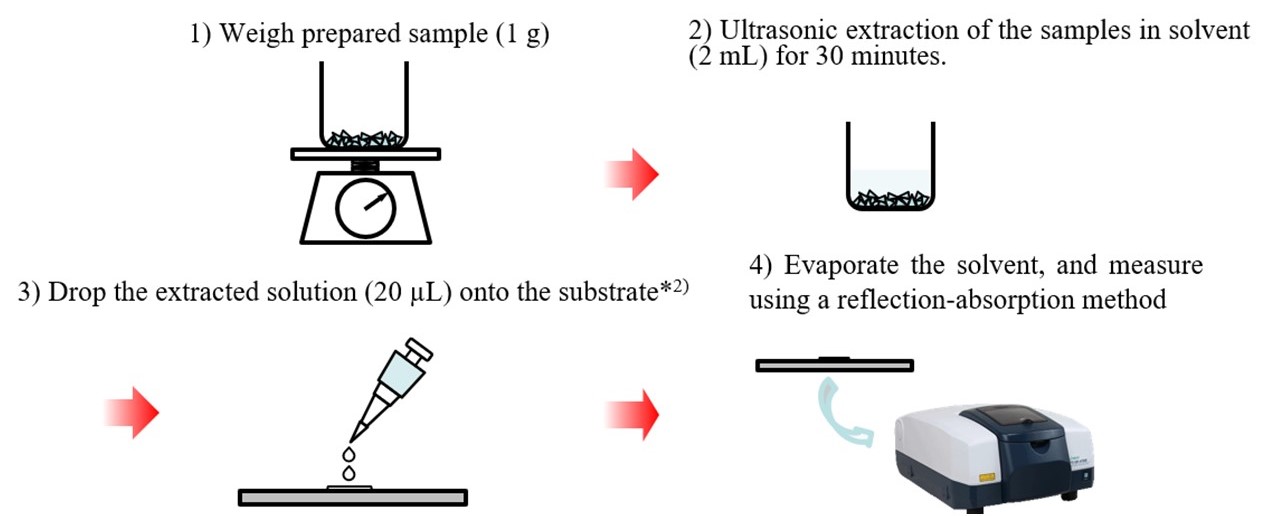
Fig.2 Measurement procedure of the extraction-condensation method
*1 If the extraction efficiency of PVC resin is 70%, in the case of resins other than PVC resin, further verification of the extraction efficiency is required.
*2 This substrate is made by Kankyo Assist Co., Ltd.
Evaluation example
The results for evaluation of phthalates concentration from a polyester polyurethane tube by extraction-condensation method is detailed below.
【Sampling/Measurement】
1) The sample was cut into small pieces of several mm with scissors and weighed (Fig.3). The total sample weight was 1002 mg.

Fig.3 Prepared sample
2) Put the sample into the mixed solvent (2 mL, acetone : hexane = 3 : 7), and use ultrasonic extraction for 30 minutes.
3) Drop the extracted solution (20 µL) onto the substrate, and evaporate the solvent.
4) Measure the thin film sample using a reflection-absorption method.

Fig.4. FT/IR-4700 with SMART-400i
Results
The spectrum on the left side in Fig.5 shows the entire spectrum, and the spectrum on the right side shows a zoomed view of key band region of phthalates (benzene ring stretching vibration). The spectra of the actual sample and standard phthalate sample (1000 ppm) are overlaid in a zoomed view. As a result, benzenesulfonamide (plasticizer) was detected, but phthalate was not detected. It means that the sample met the standard of the revised RoHS directive.
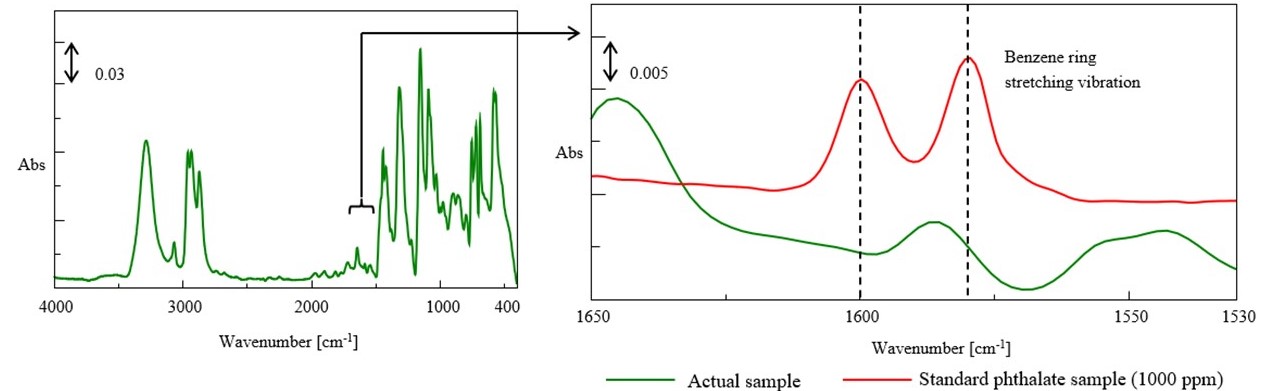
Fig.5 IR spectrum of polyester polyurethane tube (extraction-condensation method)
Conclusion
FTIR cannot be used to quantify each of the restricted phthalates, but it can be used to quantify ‘total’ phthalates. Since FTIR is much faster and easier than GC/MS and Py/TD-GC/MS, it can be used as a rapid screening techniques to reduce the burden of using the more complex and expensive MS-based techniques.