Introduction
Melamine is an organic nitrogen compound which has a triazine ring in the center of its structure. It is used as the material of melamine resin which has high thermal and water resistance and is used for mechanical strengthening. Recently, there was a case that melamine was used as an additive to diluted milk to make-up for the lack protein content, however, this milk product caused health problems. The Food and Drug Administration (FDA) has been evaluating the risk of melamine and has assigned HPLC with UV detection as one of the analysis methods of melamine in food.
In this article, the analysis results of melamine in a milk product was measured using a PDA detector.

LC-4000 HPLC system
Experimental
Experimental conditions
Column: ZORBAX RX-C8 (4.6 mmID x 150 mmL, 5 µm)
Eluent: 10mM Citric acid, 10mM Sodium-1-octanesulfonate in Water/Acetonitrile/Methanol (85/7.5/7.5)
Flow rate: 1.0 mL/min
Column temp.: 40ºC
Wavelength: 200-400 nm
Injection volume: 10 µL
Standard sample: Melamine 10 mg/mL in Water
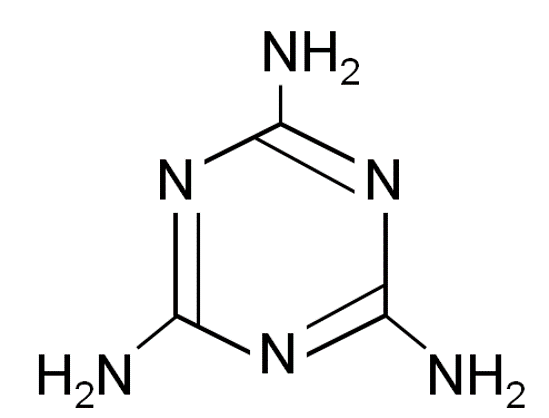
Figure 1. Structure of melamine
Keywords
FDA, Food Chemicals Codex (FCC), CFR Titile 21
Results
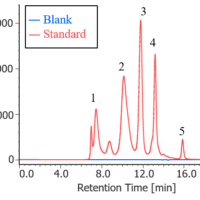
Figure 2 shows the chromatogram and contour plot of melamine.
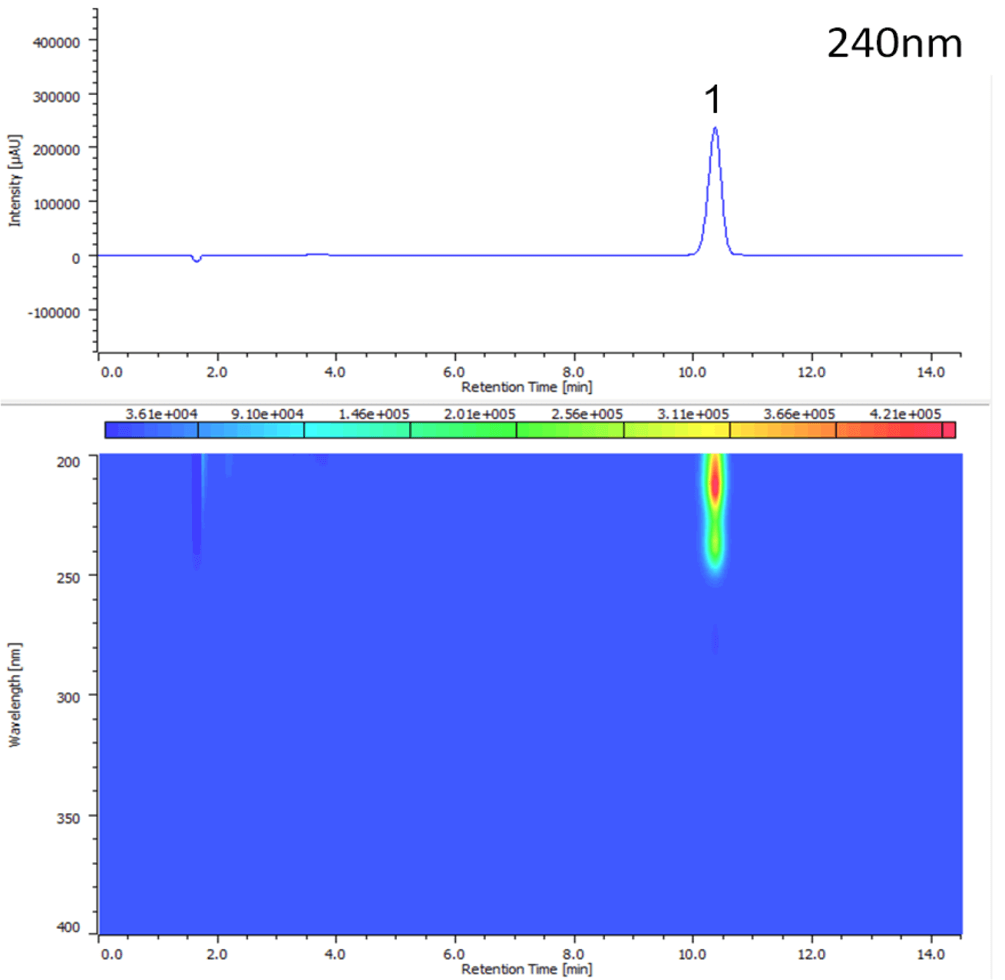
Figure 2. Chromatogram of melamine (1. Melamine)
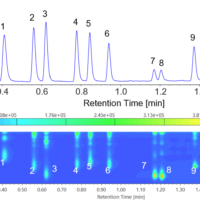
Figure 3 shows the on peak spectrum of the melamine standard sample.
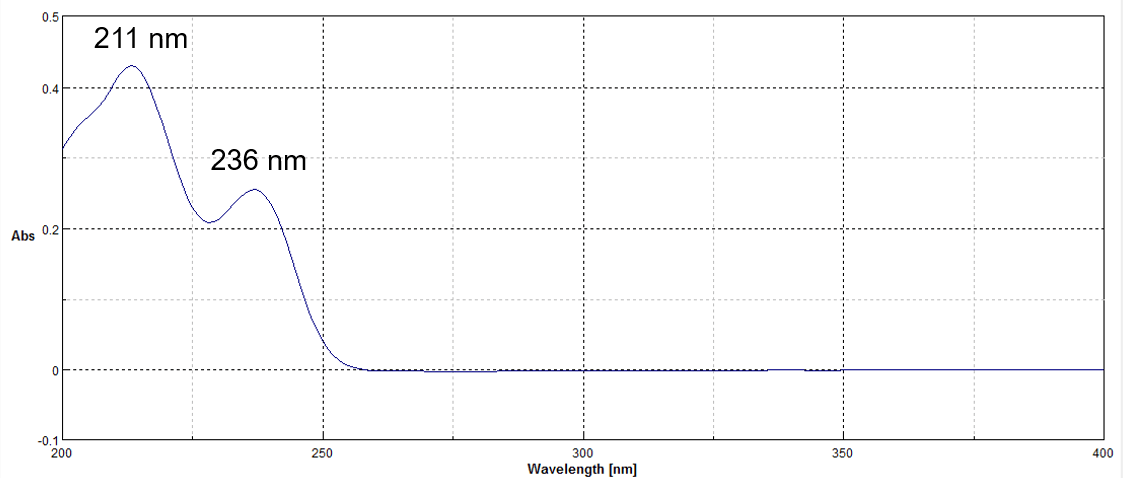
Figure 3. On peak spectrum of the melamine standard sample.
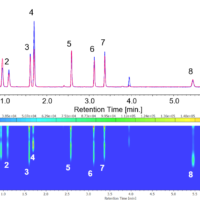
Figure 4 shows the chromatogram and on peak spectrum of milk spiked with melamine. The correlation coefficient of the spectrum of standard sample (Figure 3) and the measured one was 1.000.
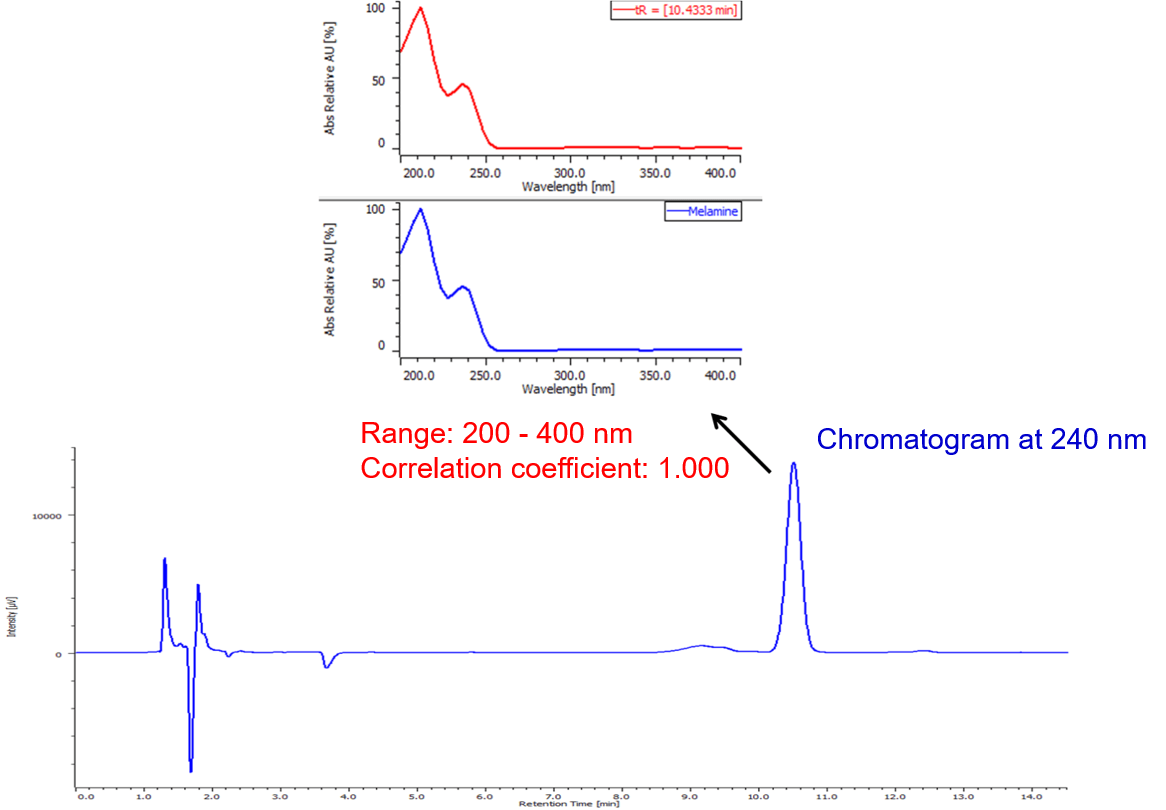
Figure. 4 Chromatogram of milk spiked with melamine.
Sample preparation: 20 µL of 100 µg/L melamine solution was added to 180 µL of milk, diluted with 1800 µL of ultrapure water and added to 200 µL of eluent. The solution was centrifuged at 5000 rpm for 10 minutes and then the supernatant was filtrated using 0.45 µm membrane filter.