Introduction
Light emission as a result of photo-excitation can occur partially in the form of fluorescence and partially as phosphorescence. Phosphorescence occurs when an intersystem crossing occurs, and electrons cannot return directly to the ground state, but instead make a transition to an intermediate state. They then relax slowly to the ground state, resulting in phosphorescence.
Fluorescence occurs on a timescale of nanoseconds, whereas phosphorescence can continue for much longer periods. When electronic transitions occur in a substance, electron pairs generally have opposite spins (Pauli’s exclusion principle). However, when an intersystem crossing occurs, the spins are the same. Since this is a forbidden state, relaxation to the ground state is slow.
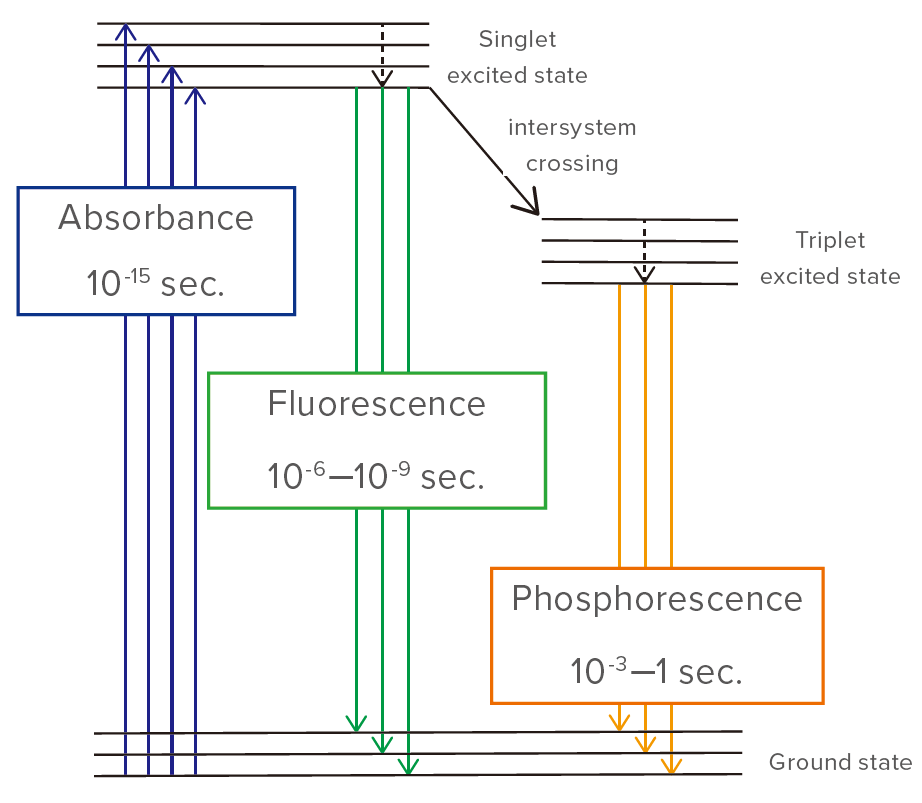
Figure 1. Phosphorescence due to intersystem crossing
When measuring phosphorescence substance by spectrofluorometer, the sample is irradiated with excitation light, and then the light after cutting the excitation light should be observed. Since fluorescence and phosphorescence are mixed while the sample is irradiated with excitation light, the phosphorescence only cannot be observed. Therefore, utilizing the property that lifetime of phosphorescence is longer than the one of fluorescence, cutting the excitation light enables to observe phosphorescence only.
JASCO’s spectrofluorometer has the shutter at the monochromator of excitation side, which enables to measure the phosphorescence. Figure 2 shows the signal of fluorescence/phosphorescence when opening/closing the shutter. When opening the shutter, the sample is irradiated with excitation light, and the observed signal is mixture of fluorescence and phosphorescence. When closing the shutter, the phosphorescence only is observed.
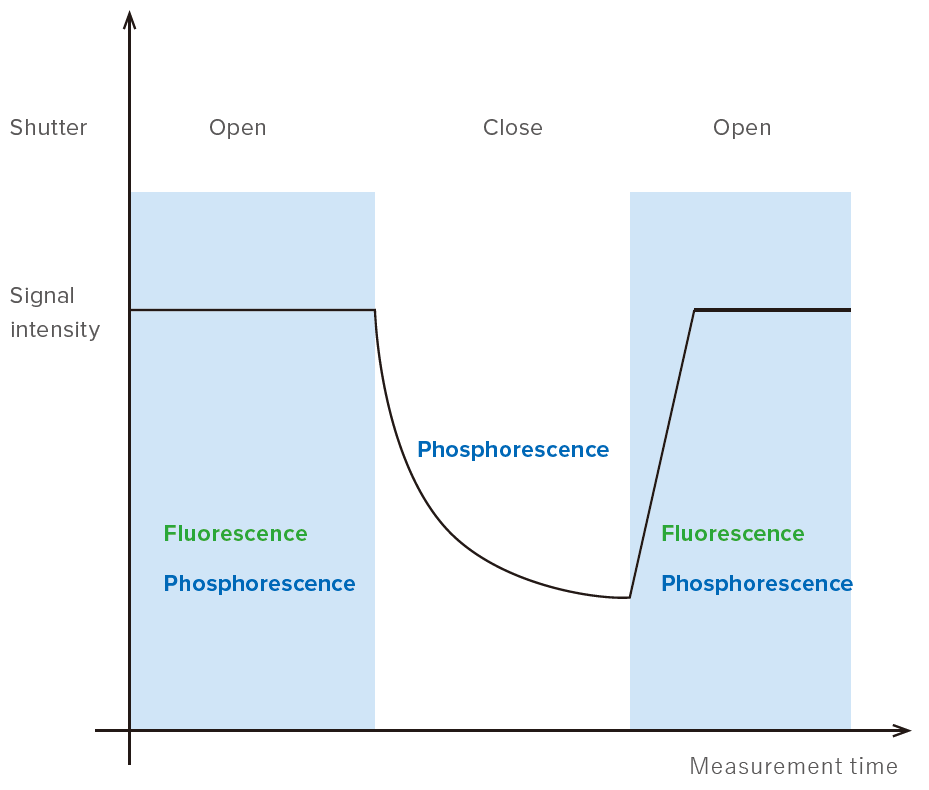
Figure 2. Observed signal when opening/closing the shutter
This section shows the evaluation example of phosphorescence.

FP-8550 Spectrofluorometer
Phosphorescence quantum yield measurement
Phosphorescent compounds are frequently used as the luminescent layer in organic electro-luminescent devices. Quantum yield measurement of phosphorescence substances is required for developing such materials. JASCO developed a new dedicated system for calculating quantum yield from the measured phosphorescence spectra at 77K. A dedicated system was evaluated by using benzophenone which is a representative phosphorescence substance. Figure 3 illustrates the measured spectra and Table 1 shows the calculation results of quantum yield (φ). The calculated phosphorescence quantum yield was 0.93 that corresponds well with the literature-based value of 0.9[1].
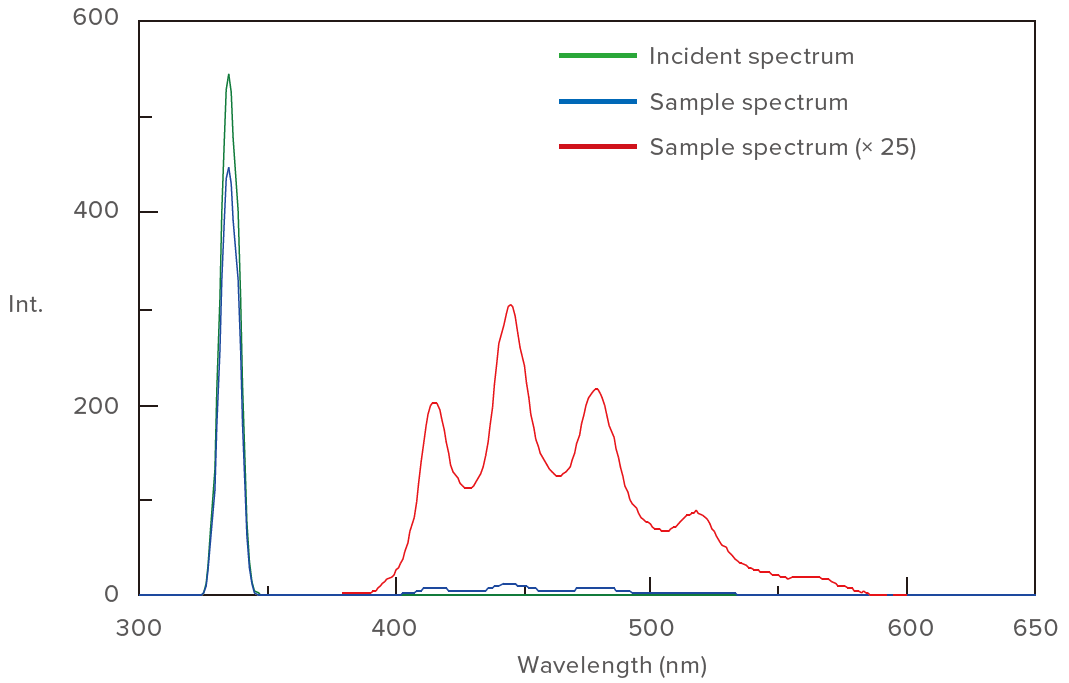
Figure 3. Spectra of benzophenone
Table 1. Phosphorescence quantum yield of benzophenone
| Int. | |
| Incident photon number: S0 | 4954.3 |
| Photon number unabsorbed by sample: S1 | 4074.0 |
| Photon number emitted by sample: S2 | 819.3 |
| Quantum yield [Measured] | 0.93 |
| Quantum yield [Literature] | 0.90 |
References
[1] The chemical society of Japan, Courses in Experimental Chemistry 3 basic physical chemistry, Maruzen ISBN: 4-621-07303-6