Introduction
Bicalutamide is a newly listed drug in the 18th edition of the Japanese Pharmacopoeia (JP) for the treatment of prostate cancer. This drug inhibits the binding of male hormones (androgens) to androgen receptors, thereby suppressing the growth of cancer cells and exerting an anti-tumor effect.
In this report, we present the results of purity and assay on bicalutamide in accordance with the Japanese Pharmacopoeia, 18th Edition.

LC-4500 series HPLC system
Experimental
<Instruments>
Pump: PU-4580
Degassing unit: DG-4580
Low pressure gradient unit: LG-4580
Autosampler: AS-4550
Column oven: CO-4060
UV detector: UV-4570
<Conditions>
Column: InertSustain C18 (4.0 mmI.D. x 250 mmL, 5 µm)
Eluent A: 0.1 % Phosphoric acid/acetonitrile (19/1)
Eluent B: Acetonitrile/0.1 % phosphoric acid (19/1)
Gradient: A/B = 92/8 (0.0 min) -> 67/33 (20.0 min) -> 50/50 (40.0 min) -> 50/50 (47.0 min) -> 92/8 (47.1 min), 1cycle; 57 min
Flow rate: 1.0 mL/min
Column temp.: 50 ºC
Preheat coil: 0.25 mmI.D. x 1000 mmL
Wavelength: 210 nm
Inj. volume: 10 µL
Standard: Bicalutamide (The dissolving and diluting solvent is a mixture of water/acetonitrile/phosphoric acid (1000/1000/1).)
<Structure>
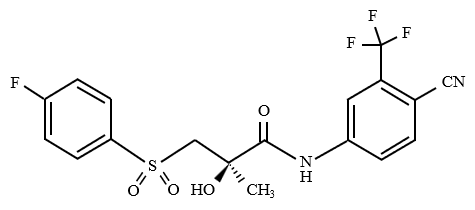
Bicalutamide
Keywords
Japanese Pharmacopoeia, bicalutamide, UV detector
Results
An overview of the system suitability for bicalutamide purity and assay is shown in Table 1.
Table 1 Overview of system suitability for bicalutamide purity and assay
| Test | Contents | |
| Purity | Detectability | When the standard solution for purity (1 mg/L) is diluted to 0.5 mg/L and the test is performed under the above operating conditions on this solution, the SN ratio for the bicalutamide peak is ≥10. |
| System performance | When the test is performed on 10 µL of the standard solution for purity under the above operating conditions, the number of theoretical plates and the bicalutamide peak symmetry factor are ≥10000 and ≤1.5, respectively. | |
| System reproducibility | When the test is repeated 6 times with 10 µL of the standard solution for purity under the above operating conditions, the relative standard deviation of the bicalutamide peak area is ≤5.0 %. | |
| Assay | System performance | When the test is performed with 10 µL of the standard assay solution (200 mg/L) under the above operating conditions, the number of theoretical plates and the bicalutamide-peak symmetry factor are ≥10000 and ≤1.5, respectively. |
| System reproducibility | When the test is repeated 6 times with 10 µL of the standard assay solution under the above operating conditions, the relative standard deviation of the bicalutamide peak area is ≤1.0%. | |
(1) Purity
Figure 1 shows the results of the detectability. For the standard solution for purity diluted to 0.5 mg/L, the SN ratio for the bicalutamide peak was 110.7, which meets the criterion of ≥10.

Fig. 1 Chromatogram of bicalutamide for detectability (0.5 mg/L)
Figure 2 shows chromatograms of bicalutamide standard solutions (n = 6), and Table 2 shows the results for peak area reproducibility based on the measured system performance and system reproducibility for purity. The number of theoretical plates was 208,383 (criterion: ≥10000), the symmetry factor was 0.98 (criterion: ≤1.5), and the relative standard deviation of the peak area was 0.12% (criterion: ≤5.0%), all of which met the specified criteria.
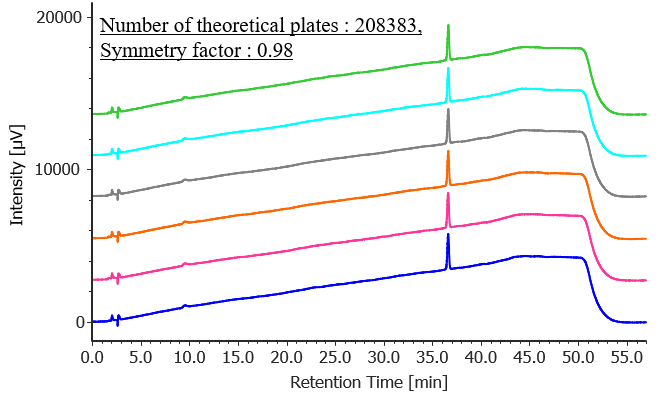
Fig. 2 Chromatogram of bicalutamide standard solutions (1 mg/L, n = 6)
Table 2 Bicalutamide peak area reproducibility (n = 6)
| Injection number | Peak area |
| 1 | 27505 |
| 2 | 27505 |
| 3 | 27521 |
| 4 | 27433 |
| 5 | 27491 |
| 6 | 27521 |
| Ave. | 27496 |
| SD | 33 |
| RSD[%] | 0.12 |
(2) Assay
Figure 3 shows chromatograms of bicalutamide standard solutions (n = 6), and Table 3 shows the peak area reproducibility based on the measured system performance and system reproducibility for the assay. The number of theoretical plates was 206,269 (criterion: ≥10000), the symmetry factor was 0.97 (criterion: ≤1.5), and the relative standard deviation of the peak area was 0.28% (criterion: ≤1.0 %), all of which all of which met the specified criteria.

Fig. 3 Chromatograms of bicalutamide standard solutions (200 mg/L, n = 6)
Table 3 Bicalutamide peak area reproducibility (n = 6)
| Injection number | Peak area |
| 1 | 5735272 |
| 2 | 5722046 |
| 3 | 5719084 |
| 4 | 5698454 |
| 5 | 5699959 |
| 6 | 5701384 |
| Ave. | 5711484 |
| SD | 15835 |
| RSD[%] | 0.28 |
Conclusion
We evaluated the system suitability for purity and assay on bicalutamide, which is newly listed in the 18th edition of the Japanese Pharmacopoeia. As shown in Table 4, all of the evaluation results met the specified criteria for the Japanese Pharmacopoeia.
Table4 Results of system suitability evaluation for bicalutamide purity and assay
| Test | Test item | Criteria | Result | Judgement | |
| Purity | Detectability | SN ratio | ≥10 | 110.7 | Passed |
| System performance | Number of theoretical plates | ≥10,000 | 208,383 | Passed | |
| Symmetry factor | ≤1.5 | 0.98 | Passed | ||
| System reproducibility | Relative standard deviation of peak area | ≤5.0% | 0.12 % | Passed | |
| Assy | System performance | Number of theoretical plates | ≥10,000 | 206,269 | Passed |
| Symmetry factor | ≤1.5 | 0.97 | Passed | ||
| System reproducibility | Relative standard deviation of peak area | ≤1.0% | 0.28 % | Passed | |